2021 Product Recalls
4,623 recalls issued in 2021.
Showing 81–100 of 4,623 recalls
Front Trunk Hood May Open Unexpectedly/FMVSS 113
If the primary latch is inadvertently released and the secondary latch is not engaged, the hood could open unexpectedly, obstructing the driver's view and increasing the risk of a crash.
Tesla Service will inspect and reposition the latch assembly as necessary, free of charge. Owner notification letters were mailed February 18, 2022. Owners may contact Tesla customer service at 1-877-798-3752. Tesla's number for this recall is SB-21-11-003.
Lidocaine Hydrochloride Topical Solution USP 4% (40 mg/mL), 50 mL glass bottles, Rx Only, Teligent Pharma Inc., Buena, NJ 08310, NDC 52565-009-50
Superpotent Drug
Telmisartan and Hydrochlorothiazide Tablets, USP, 40 mg/12.5 mg, 30 count bottles, Rx only, Manufactured by: Torrent Pharmaceuticals LTD., Indrad-382 721, India. Manufactured for: Torrent Pharma Inc., Basking Ridge, NJ 07920. NDC: 13668-159-30
Superpotent; Hydrochlorothiazide
Lidocaine Hydrochloride Topical Solution USP 4% (40 mg/mL), 50 mL glass bottles, Rx Only, Teligent Pharma Inc., Buena, NJ 08310, NDC 52565-009-50
CGMP Deviations: Lots recalled because they were manufactured at the same facility using the same components as the products that had cracked seals in caps that were found to be superpotent.
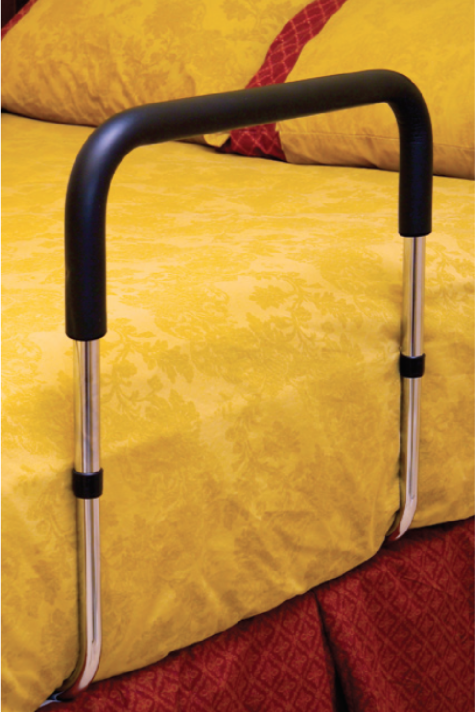
Endurance® Hand Bed Rails Recalled for Entrapment Hazard
When attached to an adult's bed, users can become entrapped within the bed rail or between the rail and the side of the mattress. This poses a serious entrapment hazard and risk of death by asphyxiation.
Consumers should immediately stop using the recalled bed rails. For consumers who own bed rails sold or imported on or after November 1, 2015, Essential Medical Supply, Inc., will provide a refund. The amount of the refund will be pro-rated based on the age of the bed rail. The company is not offering a remedy for older bed rails. Consumers with older bed rails sold between October 2006 and October 2015 should stop use, disassemble and dispose of the bed rails to prevent reuse.

Angel Line Bunk Beds with angled ladders Recalled for Entrapment Hazard
The metal hook fastening the ladder to the top bunk bed frame can move away or detach from the bed frame when the ladder is lifted, causing the gap between the ladder step and bed frame to open wider than 3.5 inches, posing serious entrapment and strangulation hazards.
Consumers should immediately stop using the recalled bunk beds, block children's access to the bunk beds and contact Angel Line to receive a free repair kit with reinforcement brackets for the ladder. Consumers who paid for the bunk beds to be assembled when purchased will receive reimbursement for help with installing the repair kit if needed.

Pearl River propane hoses used on propane torches and a Recalled for Fire Hazard
The propane hose can swell and rupture, allowing propane gas to leak, posing a fire hazard to the user.
Consumers should immediately stop using the recalled propane hoses. To obtain a free replacement hose, send a photograph of the propane hose cut in half so that it is no longer operable to the firm's e-mail along with a return mailing address to receive a free replacement hose. Once Pearl River receives confirmation of the hose cut in half, consumers can throw away the cut hose.

Carex Brand Adult Portable Bed Rails Recalled for Entrapment Hazard
When attached to an adult's bed, users can become entrapped within the bed rail or between the rail and the side of the mattress. This poses a serious entrapment hazard and risk of death by asphyxiation.
Consumers should immediately stop using the recalled bed rails and contact Compass Health Brands for a free repair kit for the Bed Support Rail or a refund for the Easy Up 2-in-1 Bed Rail.

Royal Sovereign/Royal Centurian portable air conditione Recalled for Fire Hazard
A faulty drain motor in the air conditioners can ignite the plastic enclosure of the unit, posing fire and burn hazards to consumers and risk of serious injury or death.
Consumers should immediately stop using the recalled portable air conditioners, unplug the unit, follow the instructions online at www.Recall-PAC3012.com for properly severing the electrical cord rendering the unit unusable, and contact Royal Sovereign for a pro-rated refund based on the age of the portable air conditioner. Failure to unplug the unit could result in shock, electrocution, or death.

The Captain Hang-on Treestands (2021 Batch #2M-0121) Recalled for Fall Hazard
The crimps of the plastic-coated cables can slip during use causing the standing platform to release, posing fall and injury hazards to users.
Consumers should immediately stop using the recalled treestands and contact Big Game Treestands to receive either replacement cables or to return the treestand for a full refund. Big Game Treestands is contacting all known consumers directly. To receive replacement cables, the company will set up pick-up of the original cables and delivery of the replacement cables to the consumers address. If a full refund is requested, the consumer will be asked to mail back the entire treestand using a pre-paid label at no cost to the consumer.
B-Complex, injection, 1 mL vials, Rx only, Cape Drugs 1384 Cape St. Claire Road Annapolis, MD 21409
Lack of Processing Controls
Clindamycin and Benzoyl Peroxide Gel, 1%/5% 25 g jars, Rx only, Mfg by TOLMAR Inc. Fort collins, CO 80526, NDC 0781-7263-68
Superpotent Drug
Lidocaine Hydrochloride Topical Solution USP 4% (40 mg/mL), 50 mL glass bottles, Rx Only, Manufactured by: Teligent Pharma Inc., Buena, NJ 06310, Distributed by: McKesson Corporation dba Sky Packaging, 497 Southridge Blvd, Suite 101, Memphis, TN 36141, NDC 63739-997-64.
CGMP Deviations: Lots recalled because they were manufactured at the same facility using the same components as the products that had cracked seals in caps that were found to be superpotent.
Methylcobalamin 12mg/ml injection, 1 mL vials, Rx only, Cape Drugs 1384 Cape St. Claire Road Annapolis, MD 21409
Lack of Processing Controls
Diclofenac Sodium Topical Solution, 1.5% w/w, packaged in 150 mL bottles, Rx only, Mfg: SOLA Pharmaceuticals Preferred Pharmaceuticals, Inc., NDC 68788-7707-01
Defective container: Out of specification for container integrity leading to bottles leaking.
Livia One Organic Daily Topical Spray Probiotics; 4 oz. (120 ml) UPC: 0 30915 10710 5 Recommended use: Topical: Spray affected area 2-5 times daily as needed. Ingredients: Proprietary probiotic blend in an enzyme enriched substrate: Lactobacillus-Acidophilus, L. Rhamnosus, L-Salivarius, L. Casei, L-Plantarum, Lactococcus-Lactis, Streptocuccus-Thermophilus, Bifidobacterium-Bifidum, B-Lactus, B-Infantis, B-Breve, B-Longum. Made with water and a proprietary blend of three organic grass
Probiotic products contaminated with Pseudomonas aeruginosa .
Kool-Aid cherry drink mix powder 19 oz. UPC 043000953532 19 oz. UPC 043000034163 63 oz. UPC 043000082201
potential for glass and metal in product
Country Time Lemonade drink mix powder 19 oz. UPC 043000951170 29 oz. UPC 043000951194 63 oz. UPC 043000082195 82.5 oz. UPC 043000928608 116 oz. UPC 043000075388
potential for glass and metal in product
Krispy Kreme DOUGHNUT Bites ORIGINAL GLAZED 5 1.6 OZ (45g) POUCHES NET WT. 8 OZ (227g) Manufactured under the authority of Krispy Kreme Doughnut Corporation Winston-Salem, NC 27102 UPC 0 72470 00320 1
The firm was notified by customers that the product contained metal fragments
Tang orange drink mix powder 20 oz. UPC 043000032275 58.9 oz. UPC 043000082171 72 oz. UPC 043000032268
potential for glass and metal in product