2010 Product Recalls
1,800 recalls issued in 2010.
Showing 1721–1740 of 1,800 recalls
Bausch & Lomb Millennium Phacoemulsification Needles, Needle Tapered Hub 15 Degree, REF DP8115, 6 needles per pouch, nonsterile.
The directions for use are printed with the incorrect symbols to indicate the product is packaged as sterile and nonreusable
The recalling firm issued letters dated 3/26/08 through a contracted recall notification company who issued the letters via UPS return receipt. A confirmation form was enclosed which was to be completed and returned. If you have questions, contact Customer Service at 1-800-338-2020.

Connector kits used with electronic door lock control m Recalled for Poisoning Hazard
The pin connector can fail and prevent a door from being unlocked from the inside, posing an entrapment hazard to consumers. This failure can lead to the inability to vacate a location in an emergency.
Owners of the kits should contact Stanley Convergent Security Solutions or their Sonitrol franchised dealer to schedule an appointment to have the pin connectors replaced free of charge.

Lysol Steam Cleaning Mop Recalled for Burn Hazard
Hot water mixed with Lysol can forcefully spurt out and rupture the housing unit, posing a burn hazard and a laceration hazard to consumers from the broken housing unit.
Consumers should immediately stop using the recalled mop and contact Conair to receive a full refund.
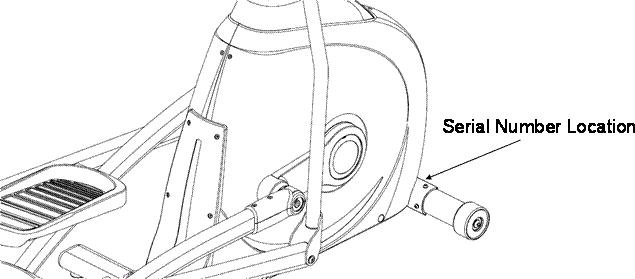
Horizon Fitness and LIVESTRONG™ Fitness Elliptical Trai Recalled for Fall Hazard
The foot pedal can become disengaged, posing a fall hazard.
Consumers should stop using the elliptical machines immediately and contact Johnson Health Tech NA to schedule a free in-home replacement of the defective pedals.

Pedrini® Pro Chop™ Professional Multipurpose Choppers Recalled for Laceration Hazard
Pieces of the chopper's metal blades can break off during use and fall into food being prepared in the chopper, posing a laceration hazard to consumers.
Consumers should immediately stop using the Pedrini® Pro Chop™ Professional Multipurpose Choppers and return it to any Sam's Club for a full refund. Sam's Club is directly contacting consumers who purchased the recalled choppers.
Opthalmic microsurgical system-AMO WHITESTAR SIGNATURE Phacoemulsification System, Product Number NGP680300. The device is a modular ophthalmic microsurgical system that facilitates both anterior segment (i.e., cataract) and posterior segment (i.e., vitreoretinal) ophthalmic surgery.
AMO initiated this field correction event after becoming aware of a trend in complaints associated with AMO Vitrectomy Cutter used in conjunction with the AMO WHITESTAR SIGNATURE Phacoemulsification System. The physician may be unable to make a cut during cataract surgery.
On April 2, 2008, AMO began distribution of an Urgent Safety Notice to all customers in the United States via overnight carrier (Federal Express). The U.S safety notice was used as a template for all global AMO communications regarding the Urgent Safety Notice , many of which required translation into local language prior to distribution. Included in this correspondence is a listing of the serial numbers of the affected products. The Notice informed customers of the problem and gave the customers modifications to the Vitrectomy Cutter Priming Instructions. In addition, the notice informed the customers that an AMO Field Service Engineer will be scheduling a site visit to modify a pressure setting on the AMO Whitestar Signature system. Contact the AMO Phaco Technical support at 1-877-AMO-4LIFE for assistance. AMO will provide updates to the FDA, including all information regarding the recall effectiveness as part of the recall progress reports.
ADJUSTABLE BRAKE PEDAL ASSEMBLIES
A REDUCTION IN BRAKING OR INTERFERENCE WITH THE THROTTLE PEDAL COULD RESULT IN A VEHICLE CRASH.
NAVISTAR IS WORKING WITH KONGSBERG PLEASE SEE KONGSBERG DEFECT REPORT 09E-064. NAVISTAR WILL RECEIVE KITS AND WILL INSPECT AND INSTALL A C-CLAMP ON THE ADJUSTABLE BRAKE PEDAL. THIS SERVICE WILL BE PERFORMED FREE OF CHARGE. THE RECALL BEGAN ON FEBRUARY 4, 2010. OWNERS MAY CONTACT NAVISTAR AT 1-800-685-6545.
ADJUSTABLE BRAKE PEDAL ASSEMBLIES
A REDUCTION IN BRAKING OR INTERFERENCE WITH THE THROTTLE PEDAL COULD RESULT IN A VEHICLE CRASH.
DAIMLER TRUCKS, THE PARENT COMPANY OF FREIGHTLINER, HAS INDICATED THAT THEY ARE CONDUCTING THE OWNERS' NOTIFICATION AND WILL BE ADMINISTERING THIS RECALL CAMPAIGN PLEASE SEE DAIMLER'S DEFECT REPORT 09V-461. DEALERS WILL RECEIVE KITS AND WILL INSPECT AND INSTALL A C-CLAMP ON THE ADJUSTABLE BRAKE PEDAL. THIS SERVICE WILL BE PERFORMED FREE OF CHARGE. THE RECALL BEGAN ON JANUARY 22, 2010. OWNERS MAY CONTACT DAIMLER TRUCKS WARRANTY CLAIMS DEPARTMENT AT 1-800-547-0712.
EP-4 Computerized Cardiac Stimulator; SJM Part Numbers: 09-1527-0000 / 09-1527-0002; EP MedSystems, Inc. 575 Route 73 North, Units D, West Berlin, NJ 08091. Indicated for use during clinical electrophysiology procedures.
Non-compliance with IEC Standard 60601-1 Clause 57.6 relative to fusing of AC input line. The neutral line is not fused. The hot line in fused. Clause 57.6 calls for both lines to be fused.
All foreign customers were sent Urgent Product Safety Information letters on March 23, 2009. Letters described units affected, the reason for action, and the actions to take to eliminate risk. It is recommended that the information in the letter be circulated to all users, risk managers and the biomedical engineering department responsible for maintaining and inspecting electrical equipment located within their institution. The letter also stated that a SJM representative will be in touch to perform the EP-4 Stimulator fuse modification. Customers are to complete and return the attachment.
Medtronic Integrated Power Console System, IP X1 REF 1898001 or REF EC300 Medtronic Xomed, Inc., 6743 Southpoint Drive North, Jacksonville, FL 32216-0980 USA. Distributed by: Medtronic Powered Surgical Solutions, 4620 North Beach Street, Fort Worth, TX 76137 USA. No difference between two referenced Consoles (REF numbers differ for internal sales tracking purposes).
An error code (Code 13) is displayed on some of the consoles when a Medtronic Stylus or EHS handpiece is connected to the IPC indicating that the handpiece has not been recognized by the console.
A Product Correction Notification was issued to each consignee on June 15, 2009. Medtronic sales representatives will visit each facility to advise consignees of the issue and to install a software upgrade that will correct the handpiece recognition issue on the console(s).
RITA Starburst XL (3-5 cm Diameter/25cm Length), Electro-surgical Device, Manufactured by: RITA Medical Systems, Inc., One Horizon Way, Manchester, GA 31816
The product may have a cracked tray which can compromise the sterility of the product.
Notification by letter of the AngioDynamics, Inc. sales force and the domestic and international distributors began on April 2, 2007. They were asked to return all affected devices to the Manchester, GA division of AngioDynamics, Inc. A reply form was attached to be completed and returned via fax to AngioDynamics.
Biomet Microplasty Elite Instruments Vanguard Slidex Femoral Block, 55 mm, Biomet Orthopedics, Inc., Warsaw, IN; REF 32-485150. A manual surgical instrument for general use; a non-powered, hand-held, or hand-manipulated device; instrument guide.
Falling component: The universal pin, which holds the sliding instrument saw guide in its slot, may loosen during surgical procedures and fall into the wound.
Consignees were notified by letter entitled "URGENT MEDICAL DEVICE REMOVAL NOTICE" dated 12/18/08 advising them of the problem and that the instruments should be removed from use and returned. For further information, please contact Biomet, Inc., by telephone on 800-348-9500.
Solar Shoulder Humeral Stem These devices are modular components of a total shoulder system. These humeral stems are intended for use with humeral heads and glenoid components in primary or revision total shoulder arthroplasty.
Stryker Orthopaedics became aware of a visual defect in certain blister packages of certain devices.
Recall notification letters were sent via Federal Express on November 21, 2008 to Stryker Branches/Agencies, Hospital Risk Management, Chief of Orthopaedics and Surgeon. The letters explained the problem and indicated that there are no risk factors. Contact Stryker Orthopaedics for additional information at 1-201-831-5718.
''Alcon Ladarwave CustomCornea Wavefront System'' Aberrometer software Version 7.91
Software error could, under specific circumstances, allow the patient's centration images and data to be stored incorrectly.
The firm mailed their 'Urgent Medical Device Safety Alert' letters to health care professtionals on July 21, 2003 along with a response form. The letter alerts the user to the problem and also how to assure that no erroneous data was saved. The letter also states that a Service Representative will be visiting to install a corrective software version 7.93. The version was completed at the start of the recall and service representative visits to install the software upgrade began approximately 7/22/03.
Stryker 100 mL AutoFuser PainPump, single site continuous, 1.0 mL/hr, 1 x 2.5" ExFen catheter, sterile, REF 0531-100-025. Infusion pump for pain medication.
Failure to infuse and potential compromised sterility: The seal on the tyvek tray cover may fail, compromising sterility, and the device may fail to infuse the drug, resulting in a loss of pain mitigation.
Stryker sent all consignees a Medical Device Recall Notification letter dated 5/14/09 and instructed to locate and destroy the recalled products and to complete and return the business reply card.
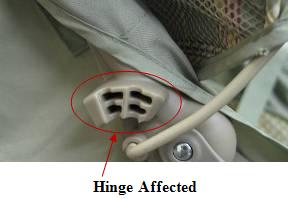
Graco's Passage™, Alano™ and Spree™ Strollers and Trave Recalled for Laceration Hazard
The hinges on the stroller's canopy pose a fingertip amputation and laceration hazard to the child when the consumer is opening or closing the canopy.
Consumers should immediately stop using the recalled strollers and contact Graco to receive a free protective cover repair kit.
BHM Reacher for Portable Ceiling Lifts; a portable ceiling lift accessory used to connect the lift to the BHM Track or KwikTrak Trolley; Manufactured by BMH Medical Inc., 2001 Tanguay Street , Magog, Quebec, Canada J1X 5Y5. The reacher comes in two models: part #700.08310 - 24'' handle length, and part #700.08320 - 36'' handle length
The carabineer may not be correctly attached to the reacher and the portable patient lift may drop.
BHM sent recall letters to Arjo''s direct accounts on 11/13/06 via first class mail, informing them that fastening the carabineer to the exterior of the closed ring portion of the reacher is contrary to the manufacturer''s instructions and is an unsafe practice. The letters informed the users that Health Canada had received notification of an incident involving a reacher marketed by a competitor, and had conducted an investigation into all similar reachers available on the Canadian market. Enclosed with the letter was a copy of the BHM Reacher User''s Guide as well as a warning sticker that visually describes the safe method for connecting the carabineer to the reacher. The accounts were requested to place the warning stickers on their reachers, review the enclosed User''s Guide in detail, and ensure that all personnel using the reacher receive refresher training on its proper use and have access to the supplied User''s Guide. Once these measures have been completed, the account was requested to complete the enclosed response form and return it to BHM by fax, e-mail or mail.

Eagle 5 Rifle Crossbows Recalled for Fire Hazard
The trigger mechanism becomes loose after 30 shots. When the safety mechanism is moved to the fire position, the crossbow will automatically discharge on its own.
Consumers should immediately stop using the recalled crossbows and contact Master Cutlery for a free replacement trigger installation. Consumers who purchased the recalled crossbow are being contacted directly.

Spine Align and Posture Perfect Ergonomic Office Chairs Recalled for Fall Hazard
The legs of the chair can break, posing fall hazard to the user.
Consumers should stop using the recalled chairs immediately and contact MooreCo for a free repair or a replacement chair.

Dorel Asia cribs Recalled for Entrapment Hazard
The drop side hardware can fail causing the drop side to detach from the crib. When the drop side detaches it creates a space in which an infant or toddler can become entrapped and suffocate or strangle. In addition, the recalled cribs can pose a serious entrapment and strangulation hazard when a slat is damaged. This can occur while the crib is in use, in storage, being put together, taken apart or reassembled; or during shipping and handling.
Consumers should immediately stop using the recalled cribs, find an alternative safe sleeping environment for your child, and contact Dorel Asia to receive a free replacement kit. Consumers should log on to www.dorel-asia.com to order the free replacement kit to prevent child entrapment in these cribs.