31 Medique Over-the-Counter drugs from the product line Recalled for Poisoning Hazard
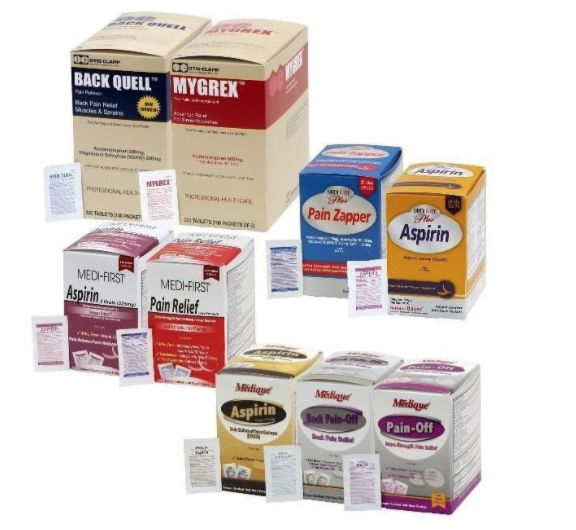
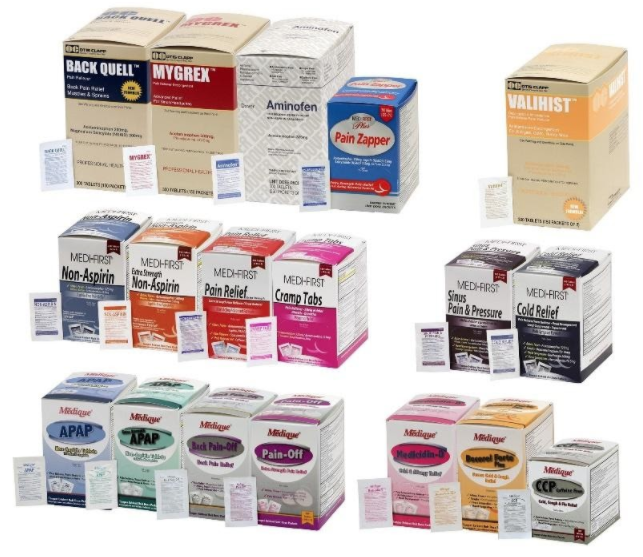

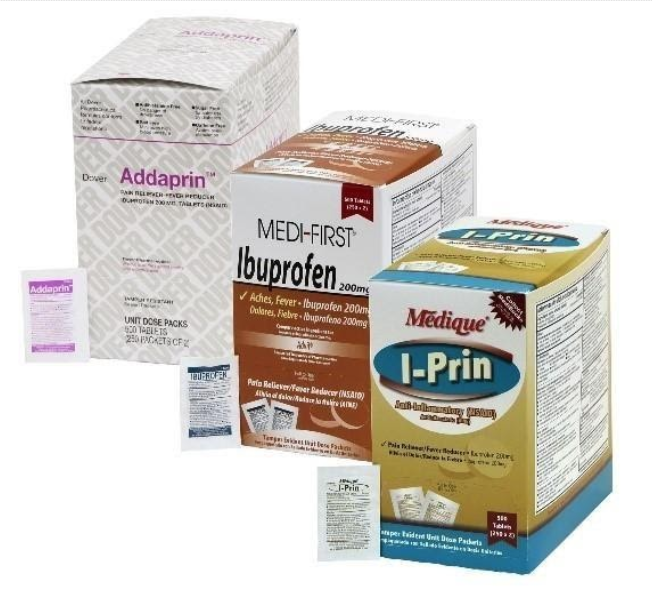


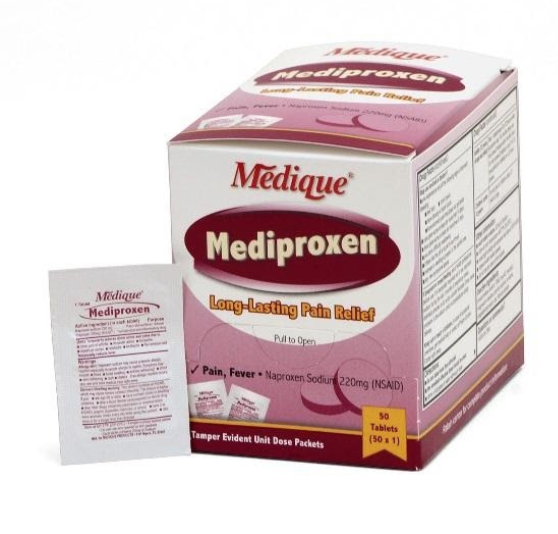
Hazard
The over-the-counter products contain regulated substances which must be in child resistant packaging when being used in the household as required by the Poison Prevention Packaging Act (PPPA). The packaging of the products is not child resistant, posing a risk of poisoning if the contents are swallowed by young children.
What You Should Do
Consumers should immediately store the recalled products in a safe location out of reach of children and contact Medique for information on how to dispose of the product and receive a full refund. All known purchasers are being notified directly.
Medique at 800-680-2474 from 8 a.m. to 7 p.m. ET, Monday through Friday, or online at www.mediqueproducts.com and click on "Recall Notice" at the bottom of the page for more information including registration access.
Get a free legal consultation about this recall.
Talk to a Lawyer →Affiliate link
Products
Injuries & Incidents
- None reported.