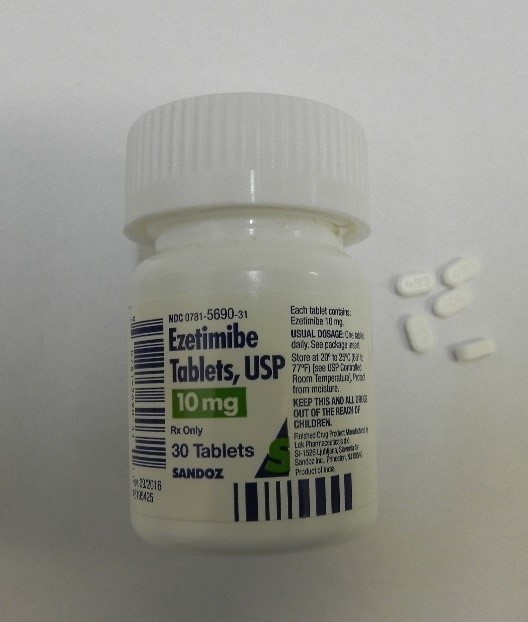
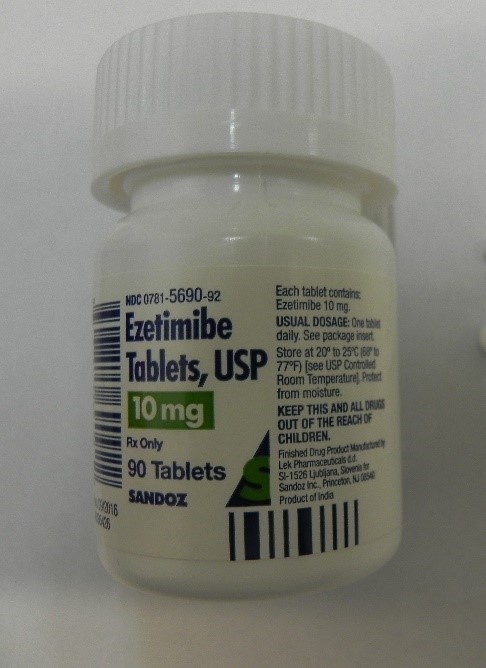
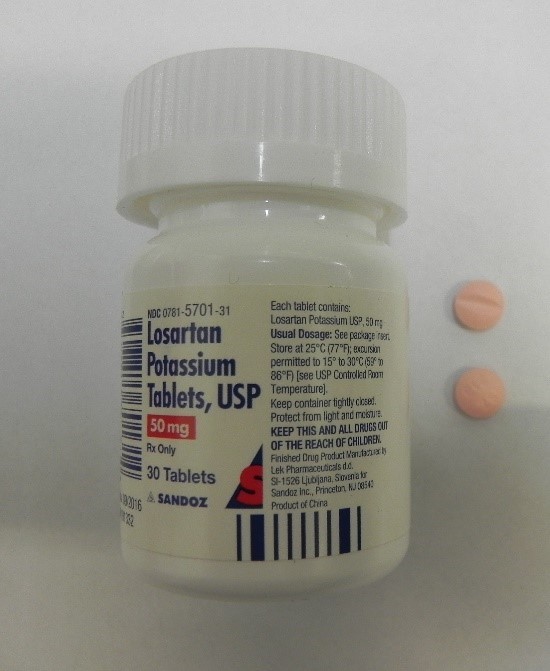
Losartan Potassium and Ezetimibe prescription drug bott Recalled for Poisoning Hazard

Hazard
The prescription drug packaging is not child resistant as required by the Poison Prevention Packaging Act, posing a poisoning risk if swallowed by children.
What You Should Do
Remedy
Consumers should immediately secure the medications to keep them out of the sight and reach of children and contact Sandoz for a free replacement child resistant bottle cap. Once the medication is secured, consumers can continue to use the medication as directed.
Consumer Contact
Sandoz at 800-525-8747 from 8:30 a.m. to 5 p.m. ET Monday to Friday or online at www.us.sandoz.com and click on “Patients and Customers” then “Product Safety Notices.”
Affected by this recall?
Get a free legal consultation about this recall.
Talk to a Lawyer →Affiliate link
Products
Losartan Potassium and Ezetimibe prescription drug bottles
Injuries & Incidents
- None Reported