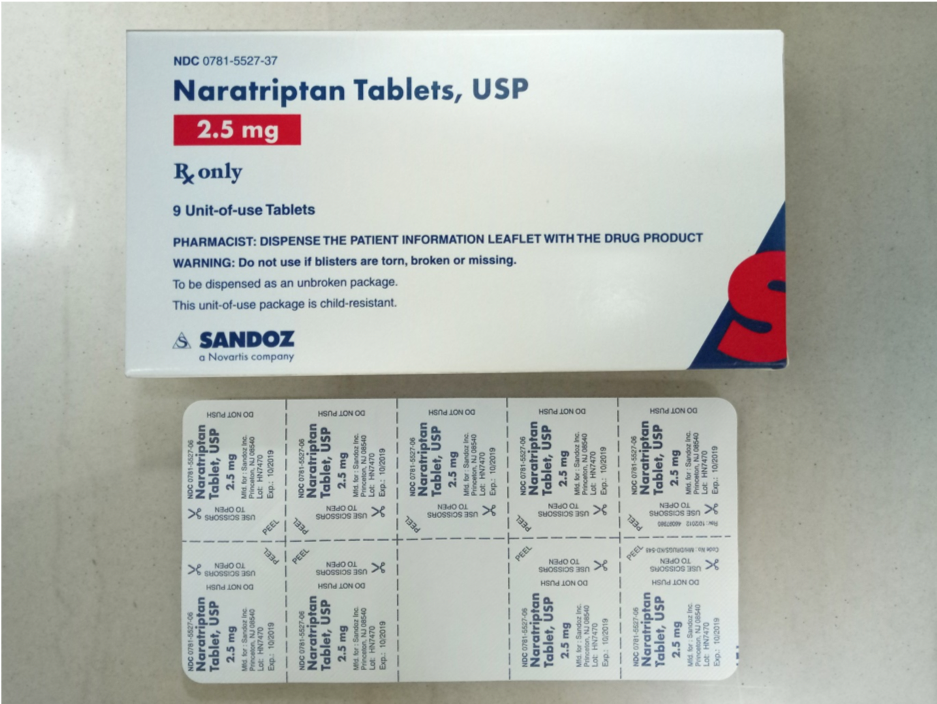
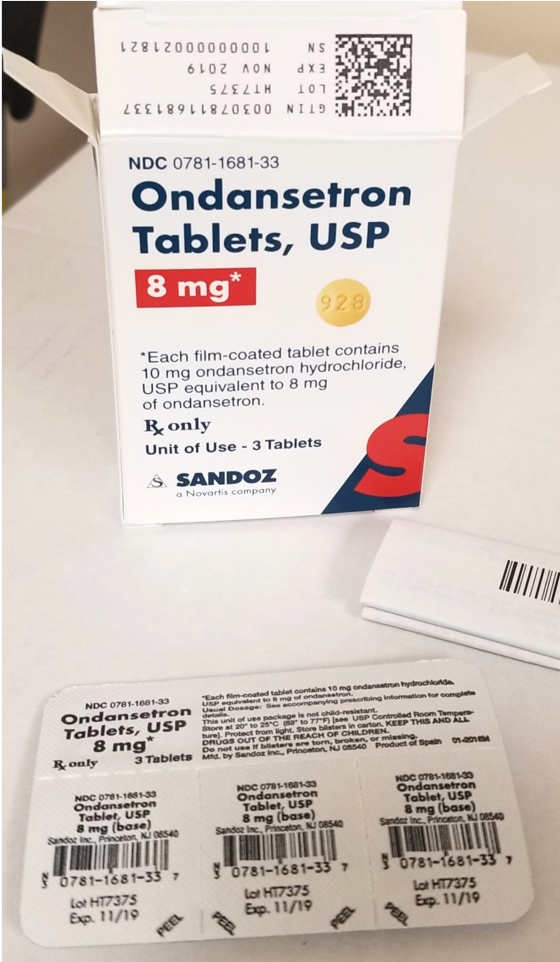
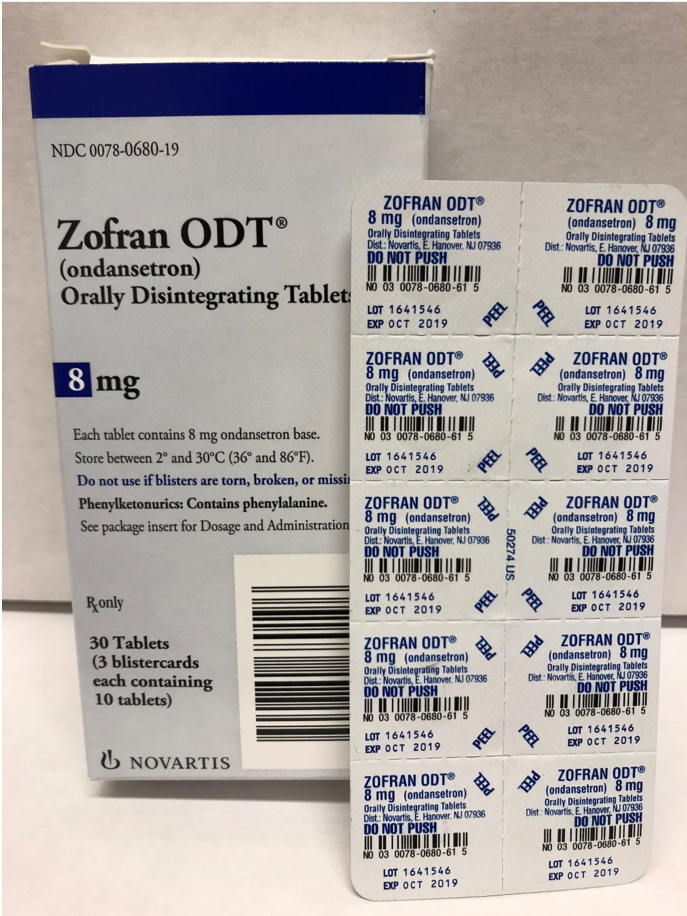
Blister packages of prescription medication Recalled for Poisoning Hazard


Hazard
The prescription drug packaging is not child resistant as required by the Poison Prevention Packaging Act, posing a poisoning risk if swallowed by children.
What You Should Do
Consumers should immediately secure the blister cards to keep them out of the sight and reach of children and contact Novartis or Sandoz for further instructions. Novartis and Sandoz advise that consumers should continue to use the medication as directed once the blister packages are secured.
Sandoz and Novartis toll-free at 888-669-6682 from Monday to Friday, 8 a.m. to 8 p.m. ET and Saturday and Sunday, 9 a.m. to 6 p.m. ET or online at www.us.sandoz.com and click on “Patients and Customers” then “Product Safety Notices,” or at www.pharma.us.novartis.com and click on banner “Novartis recalls select product blister packs.”
Get a free legal consultation about this recall.
Talk to a Lawyer →Affiliate link